research groups
Cellular-Structural Biology Lab
summary

- Binolfi, Andrés Location: CCT
Email: binolfi@ibr-conicet.gov.ar
- Piatti, Tomas
RESEARCH LINES
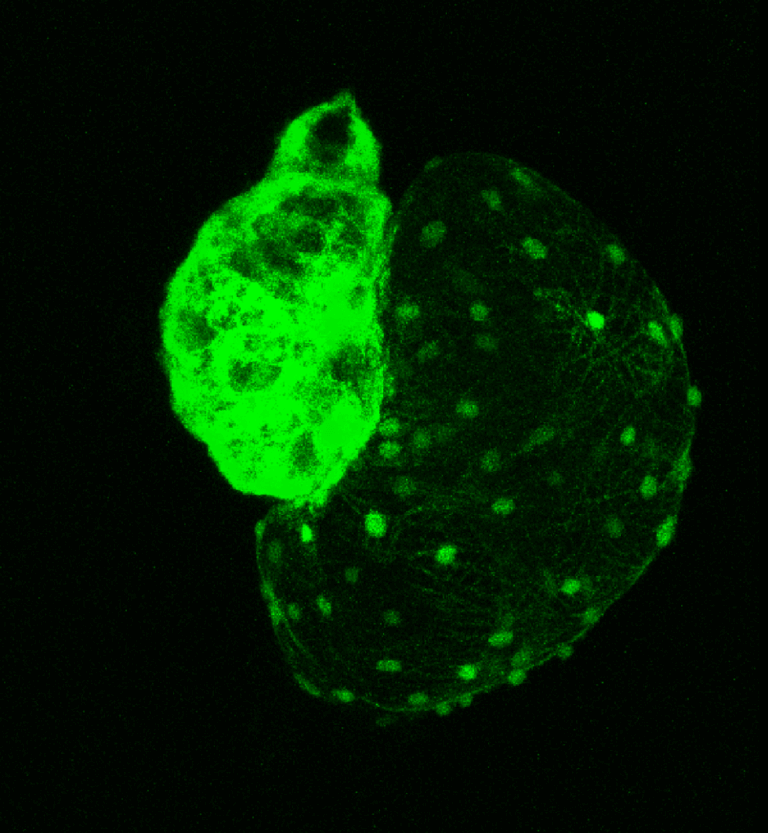
Oxidative stress and heart development
A large number of congenital heart defects (CHD) arise as a consequence of the interaction between environmental factors, infections and genetic susceptibilities. Imbalances in oxidative regulation mechanisms result in cardiac pathologies. Exposure to oxidative stress during pregnancy has been linked with CHD and a detailed knowledge of its effects on cardiac development at the morphological, molecular and cellular level constitutes the basis for the development of early detection methods and the design of novel therapeutic strategies. In this research line, headed by Dr Verónica Lombardo, we use high resolution microscopy, molecular and cell biology and NMR methodologies in zebrafish (Danio rerio) embryos to understand how oxidative stress impacts on cardiac morphogenesis and elucidate the redox signaling pathways involved in the development of congenital heart defects.
Oxidative stress, lipids and oxidative protein modifications
The increase in reactive oxygen species and the decline of enzymatic redox protective systems have been identified as risk factors for the development of neurodegenerative and cardiovascular diseases. They elicit multiple effects including protein oxidative modifications interfering with normal function and changes in the lipid composition of cell membranes, which introduce cellular stress and metabolic imbalances that promote pathological states. In a collaborative effort between our group and the group of Dr. Diego de Mendoza, we analyze the role of oxidative stress and the metabolism of unsaturated fatty acids and cholesterol in the cellular aggregation of amyloid proteins and other pathological processes. We use NMR methodologies in vitro, e in vivo, in Caenorhabditis elegans and in zebrafish, combined with protein biochemistry, microscopy and metabolomics approaches. Our initiative seeks to obtain a comprehensive high resolution picture of the effect of oxidative stress on cellular pathologies.
Images of our research lines
PUBLICATIONS AND PATENTS
Megadalton-sized dityrosine aggregates of α-Synuclein retain high degrees of structural disorder and internal dynamics.
Megadalton-sized dityrosine aggregates of α-Synuclein retain high degrees of structural disorder and internal dynamics.
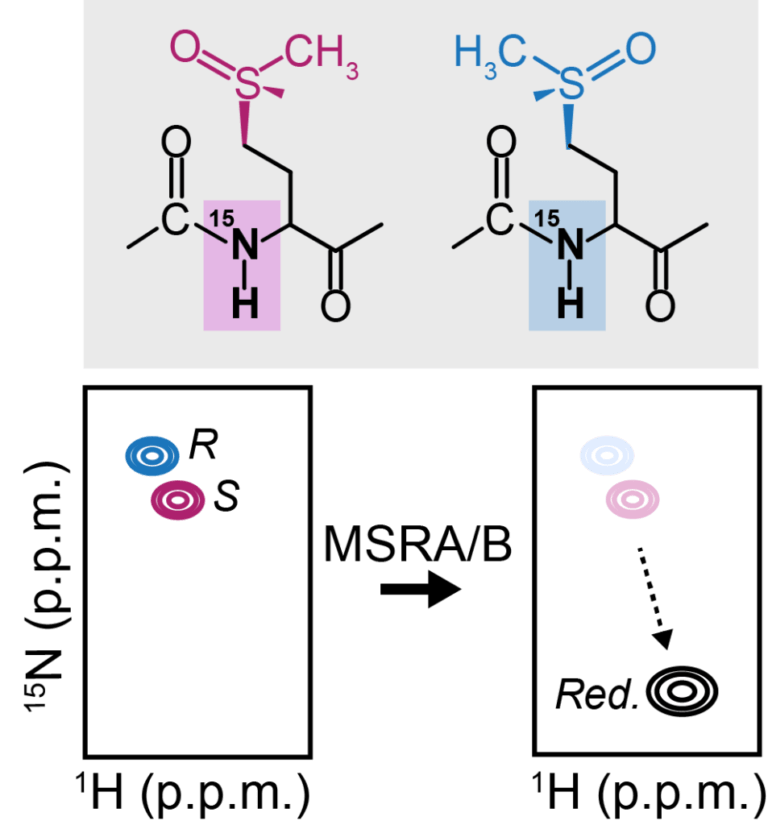
An NMR-based biosensor to measure stereo-specific methionine sulfoxide reductase (MSR) activities in vitro and in vivo
An NMR-based biosensor to measure stereo-specific methionine sulfoxide reductase (MSR) activities in vitro and in vivo
Morphogenetic control of zebrafish cardiac looping by Bmp signaling.
Morphogenetic control of zebrafish cardiac looping by Bmp signaling.
Time-resolved NMR analysis of proteolytic α-synuclein processing in vitro and in cellulo.
Time-resolved NMR analysis of proteolytic α-synuclein processing in vitro and in cellulo.
Structural disorder of monomeric α-synuclein persists in mammalian cells.
Structural disorder of monomeric α-synuclein persists in mammalian cells.
Sede CCT Rosario
Ocampo y Esmeralda, Predio CONICET-Rosario
2000 Rosario, Santa Fe, Argentina
Tel. 54-341-4237070 / 4237500 / 4237200
Sede Facultad de Ciencias Bioquímicas y Farmacéuticas
Universidad Nacional de Rosario - Suipacha 531
2000 Rosario, Santa Fe, Argentina
Tel. +54 341 4350596 / 4350661 / 4351235
💧🔬Investigadoras del IBR/Conicet/UNR trabajan en biosensores y estrategias biológicas para detectar contaminantes y mejorar el tratamiento de agua en localidades de Santa Fe.

